 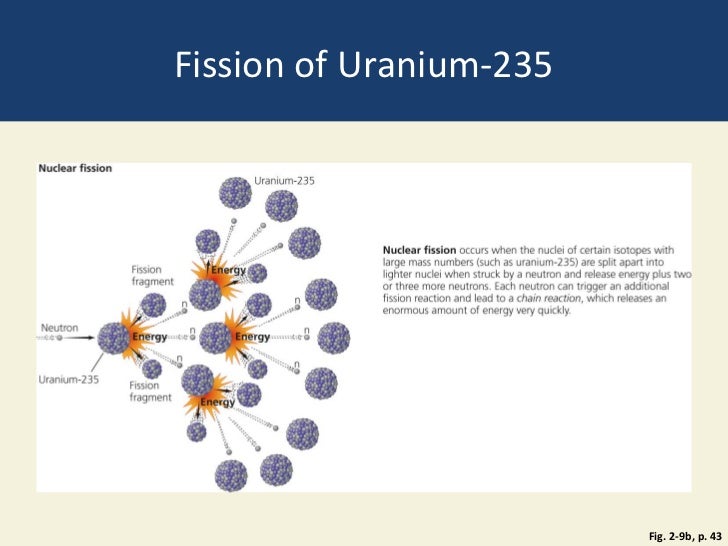
Uranium-235 is obtained from natural uranium through the process of enrichment. Source (Table 1): National Library of Medicine The relative concentrations of the three isotopes in natural uranium are: Isotope The isotope is used as fuel for nuclear power reactors and for building nuclear weapons. Uranium-235 is also the most sought-after of uranium’s three isotopes. (Fissile material is any substance capable of sustaining a nuclear fission chain reaction.) Uranium-238 is the most abundant of the three isotopes, while 235U is the only naturally occurring fissile material. Uranium occurs in nature as a mixture of three major radioactive isotopes 238U, 235U, and 234U. How much uranium can you eat? What is uranium? Uranium is element 92 on the periodic table. Calories released by 235U compared with the heating value of coal Calories released by 235U compared with TNT How many calories were in the Hiroshima and Nagasaki bombs? How many calories are released per atom of uranium-235 fissioned?įun Facts: 1g of 235U releases 20 billion calories of energy How many calories are released when 1g of uranium-235 undergoes fission How to calculate calories equal to 1g of “missing mass” when 1kg of uranium undergoes fission How many calories are in a gram of uranium?Ĭalories in nutrition vs. We also offer in-depth answers if you’ve been wondering whether you can eat uranium and what happens if you eat the metallic element: Will eating the metal give you lots of calories and make you fat? This article shows you how to use the Einstein equation E=mc 2 to calculate the number of calories released by 1g of uranium undergoing fission. Similarly, the amount of energy released when 1 kg (2.2lbs) of uranium-235 undergoes complete fission is approximately 20 billion kilocalories. Nutritional Value Of Cheesecake – The Best In-depth Guideīased on Einstein’s mass-energy equivalence principle, 1g of uranium releases 20 billion calories when it undergoes complete fission. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
